EN 14561:2006
Quantitative carrier test for the evaluation of bactericidal activity of instrument disinfectants intended for use in the medical area.
EN 14561:2006 is a mandatory phase 2 step 2 carrier test for instrument disinfectants intended for use in the medical area. The test evaluates the efficacy of the product against bacteria.
Test Conditions
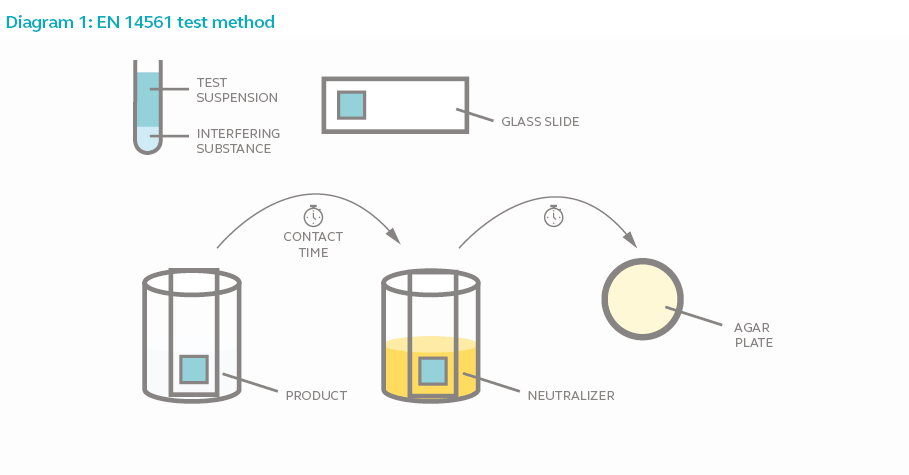
The standard refers to the parameters to be observed when testing products intended for instrument disinfection. This includes the test microorganism, test temperature, carrier, contact time and interfering substance.
- Test microorganism refers to the mandatory list of microbes that must be used in the test to determine the antimicrobial activity of the product. The mandatory microorganisms are assumed to represent all microbes in its group.
- Test temperature refers to the temperature in which the test must be conducted. The general assumption is that disinfectants are less effective in low temperatures compared to higher temperatures.
- Carrier refers to the material used in the test to simulate a contaminated instrument in the medical area. This test uses glass slide as a carrier.
- Contact time refers to the minimum duration a product must remain in contact with the microbes for the product to be effective.
- Interfering substance refers to substance used in the test to simulate the possible contaminants that co-occur with microbes in the actual environment. In a hospital for example, hepatitis B virus contamination might also contain blood and other bodily fluids which may make disinfection challenging especially if it’s dried matter.
Refer to the table below for the minimum test requirement.

Test Method
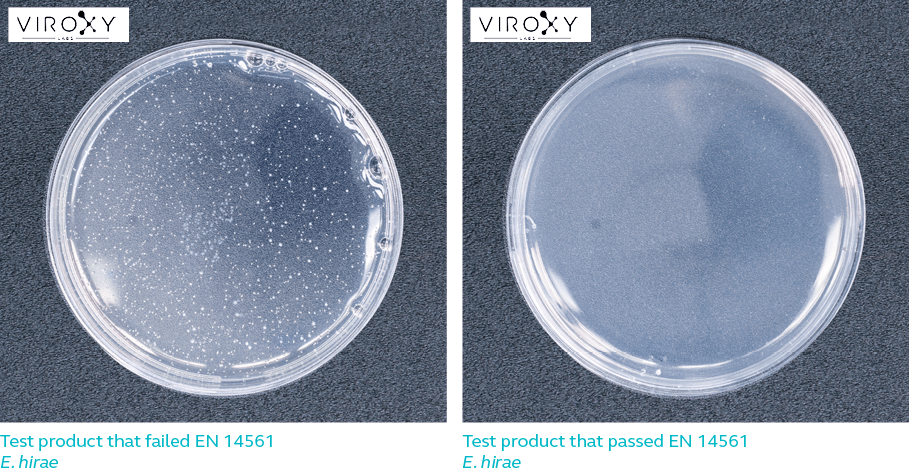
In a carrier test, the test microorganism is added to the selected interfering substance and applied onto a glass slide. The glass slide represents an actual instrument or carrier of microorganism. The glass slide is then left to air-dry to simulate instrument surface or carrier of microorganism. Next, the carrier is submerged in the test disinfectant for the duration of the contact time. The carrier is then transferred into a neutralizer solution to prevent continued bactericidal activity. A sample of the neutralizer solution is then acquired, plated and incubated for 2 days. The number of test microorganism recovered is compared to the microorganism recovered from the control sample (where test microorganism is exposed to water instead of disinfectant) to determine if the test product is effective.
Log Reduction
Log reduction refers to the extent to which a product is capable of reducing the number of microbes. For example, 4-log reduction means the number of microorganisms on a surface has been reduced by 10 000 times. A product that is 99.9% effective against a certain microbe is said to have achieved 3-log reduction against that microbe.
For a product to pass EN 14561, it must be able to achieve 5-log reduction against the respective test microorganisms listed in Table 1. In other words, the product must be able to kill 99.999% bacteria while meeting all the other requirements of the European standard.