EN 1656:2019
Quantitative suspension test for the evaluation of bactericidal activity of chemical disinfectants and antiseptics used in the veterinary area.
EN 1656 is a phase 2 step 1 suspension test for disinfectants intended for use in the veterinary area. The test evaluates the efficacy of the product against bacteria.
Test Conditions
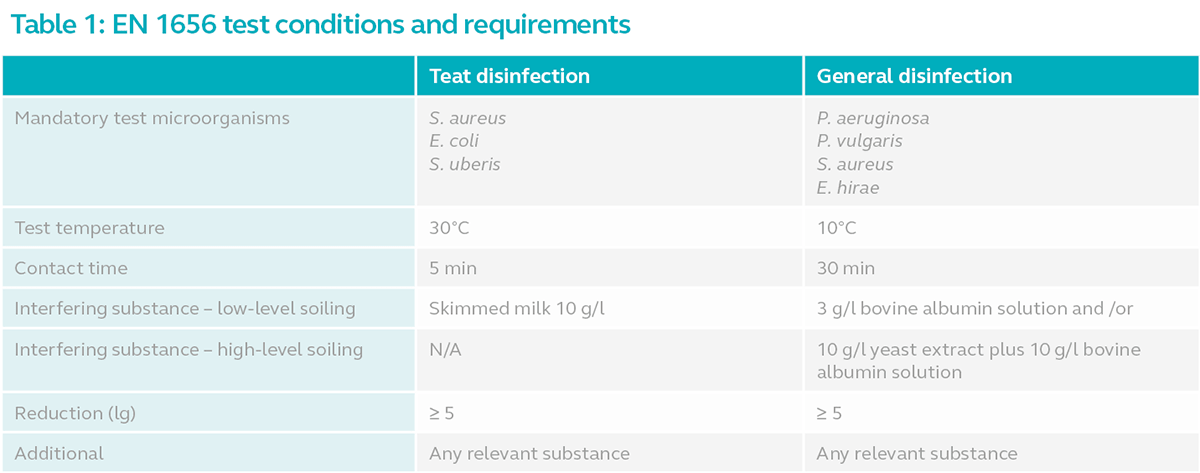
The standard refers to the parameters to be observed when testing products for teat disinfection and general disinfection. This includes the test microorganism, test temperature, contact time and interfering substance.
- Test microorganism refers to the mandatory list of microbes that must be used in the test to determine the antimicrobial activity of the product. The mandatory microorganisms are assumed to represent all microbes in its group.
- Test temperature refers to the temperature in which the test must be conducted. The general assumption is that disinfectants are less effective in low temperatures compared to higher temperatures.
- Contact time refers to the minimum duration a product must remain in contact with the microbes for the product to be effective.
- Interfering substance refers to substance used in the test to simulate the possible contaminants that co-occur with microbes in the actual environment.
Refer to the table below for the minimum and additional test conditions.
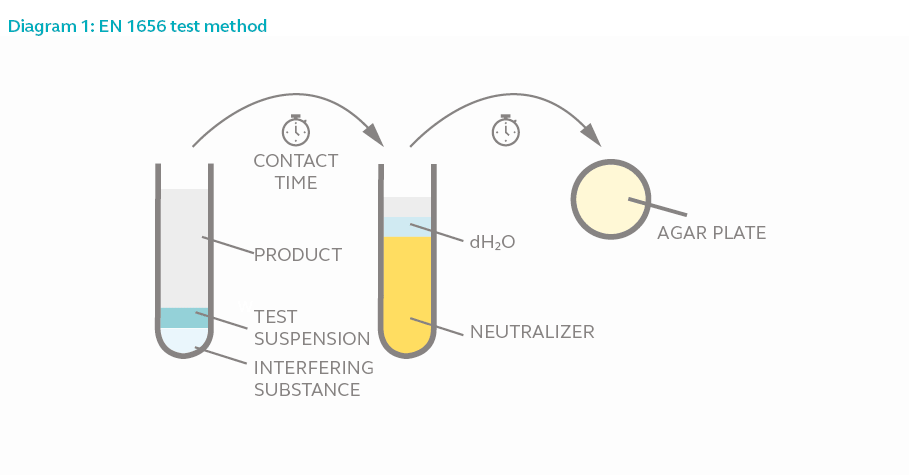
Test Method
In a phase 2 step 1 suspension test, 8 parts of the test product is added to 1-part test microorganism and 1-part interfering substance. The mixture is allowed to interact for the duration of the contact time. One part of the mixture is added to 8 parts of neutralizer and 1-part water for 5 minutes to halt bactericidal activity. The final mixture is then acquired and incubated for 2 days to allow surviving bacteria (if any) to proliferate. The bacterial colony is counted and compared against the original culture size.
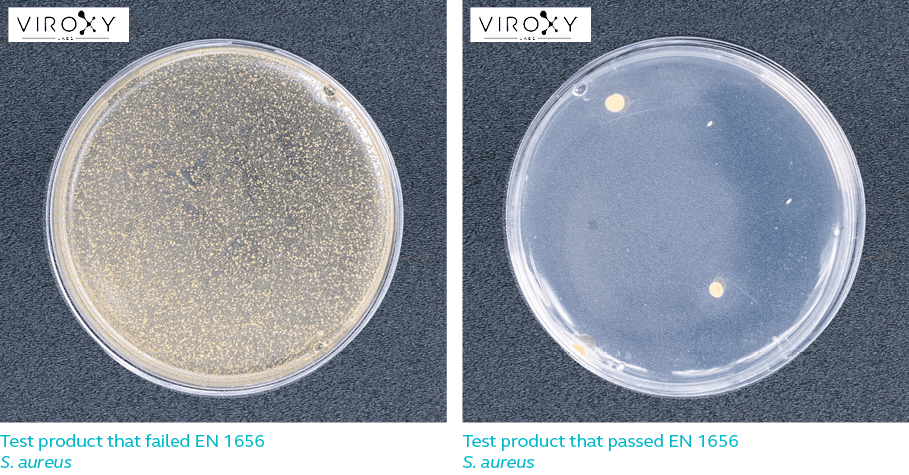
Log Reduction
Log reduction refers to the extent to which a product is capable of reducing the number of microbes. For example, 4-log reduction means the number of microorganisms on a surface has been reduced by 10 000 times. A product that is 99.9% effective against a certain microbe is said to have achieved 3-log reduction against that microbe.
For a product to pass EN 1656, it must be able to achieve 5-log reduction against the respective test microorganisms listed in Table 1. In other words, the product must be able to kill 99.999% bacteria while meeting all the other requirements of the European standard.